- Blog
- Nintendo ds emulator for mac os x
- Cardiosoft Software
- Steam racing games for mac
- Lan emulator for mac and windows
- Photo booth software for mac
- Sendblaster pro 4 serial key
- Higurashi daybreak pc
- Nintendo 2ds emulator mac
- Download film keramat full movie ganool
- Mac os x playstation 2 emulator
- Mount and blade with fire and sword crack 1-143 download
- Android emulator on mac os x slow
- Cartina geografica italia politico pdf download
- Total validator pro crack

CARDIOSOFT SOFTWARE MANUAL
Therefore, carefully read the manual once in its entirety. Please note that information pertinent to several chapters is given only once. Close observance of the information given in the manual is a prerequisite for proper equipment performance and correct operation and ensures patient and operator safety. It should be available to the equipment operator at all times.
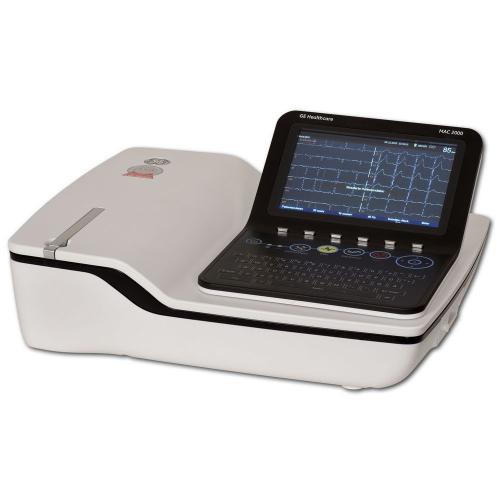

This manual is an integral part of the equipment. The CE marking covers only the accessories listed in the "Order Information" chapter. The radio-interference emitted by this product is within the limits specified in CISPR11/EN 55011, class B. The product fulfills the requirements of the standard EN/IEC 60601-1 "Medical Electrical Equipment, Part 1: General Requirements for Basic Safety and Essential Performance" as well as the electromagnetic immunity requirements of the standard EN/IEC 6 "Medical electrical equipment – Collateral standard: Electromagnetic compatibility – Requirements and tests" and applicable amendments. The device fulfills the requirements of the Directive 2011/65/EU of the European Parliament and of the Council. It has an internal power source and is an MDD class IIa device. The product TONOPORT V bears the CE marking CE-0482 (notified body MEDCERT GmbH) indicating its compliance with the provisions of the Council Directive 93/42/EEC about medical devices (including amendment 2007/47/EC) and fulfills the essential requirements of Annex I of this directive.
- Blog
- Nintendo ds emulator for mac os x
- Cardiosoft Software
- Steam racing games for mac
- Lan emulator for mac and windows
- Photo booth software for mac
- Sendblaster pro 4 serial key
- Higurashi daybreak pc
- Nintendo 2ds emulator mac
- Download film keramat full movie ganool
- Mac os x playstation 2 emulator
- Mount and blade with fire and sword crack 1-143 download
- Android emulator on mac os x slow
- Cartina geografica italia politico pdf download
- Total validator pro crack
